Artículos SCI
2023
2023
Threads of memory: Reviving the ornament of a dead child at the Neolithic village of Ba`ja (Jordan)
Alarashi, H et al. [Aviles, MA]Plos One, 18 (2023) DOI: 10.1371/journal.pone.0288075
Abstract
In 2018, a well-constructed cist-type grave was discovered at Ba`ja, a Neolithic village (7,400-6,800 BCE) in Southern Jordan. Underneath multiple grave layers, an 8-year-old child was buried in a fetal position. Over 2,500 beads were found on the chest and neck, along with a double perforated stone pendant and a delicately engraved mother-of-pearl ring discovered among the concentration of beads. The first was found behind the neck, and the second on the chest. The meticulous documentation of the bead distribution indicated that the assemblage was a composite ornament that had gradually collapsed, partly due to the burying position. Our aim was to challenge time degradation and to reimagine the initial composition in order to best explore the significance of this symbolic category of material culture, not as mere group of beads, but as an ornamental creation with further aesthetic, artisanal and socioeconomic implications. The reconstruction results exceeded our expectations as it revealed an imposing multi-row necklace of complex structure and attractive design. Through multiple lines of evidence, we suggest that the necklace was created at Ba`ja, although significant parts of beads were made from exotic shells and stones, including fossil amber, an unprecedented material never attested before for this period. The retrieval of such an ornament from life and its attribution to a young dead child highlights the significant social status of this individual. Beyond the symbolic functions related to identity, the necklace is believed to have played a key role in performing the inhumation rituals, understood as a public event gathering families, relatives, and people from other villages. In this sense, the necklace is not seen as belonging completely to the realm of death but rather to the world of the living, materializing a collective memory and shared moments of emotions and social cohesion.
Agosto, 2023 · DOI: 10.1371/journal.pone.0288075
Química de Superficies y Catálisis
Hydrogen production by catalytic aqueous-phase reforming of waste biomass: a review
González-Arias, J; Zhang, Z; Reina, TR; Odriozola, JAEnvironmental Chemistry Letters, 21 (2023) 3089-3104 DOI: 10.1007/s10311-023-01643-w
Abstract
The rising adverse effects of climate change call for a rapid shift to low-carbon energy and reducing our dependence on fossil fuels. For that, biorefineries appear as promising alternatives to produce energy, chemicals, and fuels using biomass and waste as raw materials. Here, we review catalytic aqueous-phase reforming to convert biomass and organic waste carbohydrates into renewable hydrogen, with focus on reforming basics; catalyst design; reforming of model compounds, wastewater and biomass; economics and life cycle assessment. We found that platinum and palladium are technically highly effective, yet their high price may limit upscaling. Alternatively, addition of tin to nickel gives acceptable results and improves hydrogen selectivity from 35 to 90%. We observed that hydrogen production decreases from 14% for crude glycerol to 2% for pure glycerol, thus highlighting the need to do experiments with real wastewater. The rare experiments on real wastewater from brewery, juice, tuna, and cheese industries have given hydrogen production rates of up to 149.7 mg/L. Aqueous-phase reforming could be shortly competitive with prices around 3-6 USD per kg of hydrogen, which are nearing the current market prices of 2-3 USD per kg.
Agosto, 2023 · DOI: 10.1007/s10311-023-01643-w
Materiales Ópticos Multifuncionales
Inkjet-Printed and Nanopatterned Photonic Phosphor Motifs with Strongly Polarized and Directional Light-Emission
Cabello-Olmo, E; Romero, M; Kainz, M; Bernroitner, A; Kopp, S; Muhlberger, M; Lozano, G; Miguez, HAdvanced Functional Materials, (2023) 2305907 DOI: 10.1002/adfm.202305907
Abstract
Herein a versatile and scalable method to prepare periodically corrugated nanophosphor surface patterns displaying strongly polarized and directional visible light emission is demonstrated. A combination of inkjet printing and soft lithography techniques is employed to obtain arbitrarily shaped light emitting motifs. Such predesigned luminescent drawings, in which the polarization and angular properties of the emitted light are determined and finely tuned through the surface relief, can be used as anti-counterfeiting labels, as these two specific optical features provide additional means to identify any unauthorized or forged copy of the protected item. The potential of this approach is exemplified by processing a self-standing photoluminescent quick response code whose emission is both polarized and directionally beamed. Physical insight of the mechanism behind the directional out-coupled photoluminescence observed is provided by finite-difference time-domain calculations.
Agosto, 2023 · DOI: 10.1002/adfm.202305907
Materiales Semiconductores para la Sostenibilidad
Surfactant-Dependent Bulk Scale Mechanochemical Synthesis of CsPbBr3 Nanocrystals for Plastic Scintillator-Based X-ray Imaging
Ghosh, J; O'Neill, J; Masteghin, MG; Braddock, I; Crean, C; Dorey, R; Salway, H; Anaya, M; Reiss, J; Wolfe, D; Sellin, PACS Applied Nano Materials, DOI: 10.1021/acsanm.3c02531
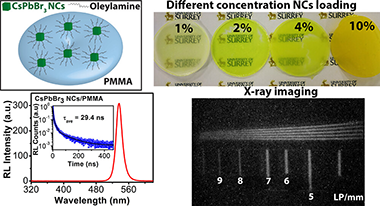
Abstract
We report a facile, solvent-free surfactant-dependentmechanochemicalsynthesis of highly luminescent CsPbBr3 nanocrystals (NCs)and study their scintillation properties. A small amount of surfactantoleylamine (OAM) plays an important role in the two-step ball millingmethod to control the size and emission properties of the NCs. Thesolid-state synthesized perovskite NCs exhibit a high photoluminescencequantum yield (PLQY) of up to 88% with excellent stability. CsPbBr3 NCs capped with different amounts of surfactant were dispersedin toluene and mixed with polymethyl methacrylate (PMMA) polymer andcast into scintillator discs. With increasing concentration of OAMduring synthesis, the PL yield of CsPbBr3/PMMA nanocompositewas increased, which is attributed to reduced NC aggregation and PLquenching. We also varied the perovskite loading concentration inthe nanocomposite and studied the resulting emission properties. Themost intense PL emission was observed from the 2% perovskite-loadeddisc, while the 10% loaded disc exhibited the highest radioluminescence(RL) emission from 50 kV X-rays. The strong RL yield may be attributedto the deep penetration of X-rays into the composite, combined withthe large interaction cross-section of the X-rays with the high-Zatoms within the NCs. The nanocomposite disc shows an intense RL emissionpeak centered at 536 nm and a fast RL decay time of 29.4 ns. Further,we have demonstrated the X-ray imaging performance of a 10% CsPbBr3 NC-loaded nanocomposite disc.
Agosto, 2023 · DOI: 10.1021/acsanm.3c02531
Química de Superficies y Catálisis
H2-rich syngas production from biogas reforming: Overcoming coking and sintering using bimetallic Ni-based catalysts
Carrasco-Ruiz, S; Zhang, Q; Gándara-Loe, J; Pastor-Pérez, L; Odriozola, JA; Reina, TR; Bobadilla, LFInternational Journal of Hydrogen Energy, 48 (2023) 72 DOI: 10.1016/j.ijhydene.2023.03.301
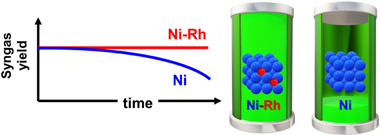
Abstract
Dry reforming of methane is a very appealing catalytic route biogas (mainly composed by greenhouse gases: carbon dioxide and methane) conversion into added value syngas, which could be further upgraded to produce liquid fuels and added value chemicals. However, the major culprits of this reaction are coking and active phase sintering that result in catalysts deactivation. Herein we have developed a highly stable bimetallic Ni–Rh catalyst supported on mixed CeO2–Al2O3 oxide using low-noble metal loadings. The addition of small amounts of rhodium to nickel catalysts prevents coke formation and improves sintering resistance, achieving high conversions over extended reaction times hence resulting in promising catalysts for biogas upgrading.
Agosto, 2023 · DOI: 10.1016/j.ijhydene.2023.03.301
Materiales Ópticos Multifuncionales
Measurement principles for quantum spectroscopy of molecular materials with entangled photons
Moretti, L; Rojas-Gatjens, E; Uboldi, L; Tiede, DO; Kumar, EJ; Trovatello, C; Preda, F; Perri, A; Manzoni, C; Cerullo, G; Kandada, ARSJournal of Chemical Physics, 159 (2023) 084201 DOI: 10.1063/5.0156598
Abstract
Nonlinear spectroscopy with quantum entangled photons is an emerging field of research that holds the promise to achieve superior signal-to-noise ratio and effectively isolate many-body interactions. Photon sources used for this purpose, however, lack the frequency tunability and spectral bandwidth demanded by contemporary molecular materials. Here, we present design strategies for efficient spontaneous parametric downconversion to generate biphoton states with adequate spectral bandwidth and at visible wavelengths. Importantly, we demonstrate, by suitable design of the nonlinear optical interaction, the scope to engineer the degree of spectral correlations between the photons of the pair. We also present an experimental methodology to effectively characterize such spectral correlations. Importantly, we believe that such a characterization tool can be effectively adapted as a spectroscopy platform to optically probe system-bath interactions in materials.
Agosto, 2023 · DOI: 10.1063/5.0156598
Reactividad de Sólidos
Touch-free reactive flash sintering of dense strontium hexaferrite permanent magnet
Jalali, SIA; Manchon-Gordon, AF; Chacartegui, R; Sanchez-Jimenez, PE; Blazquez, JS; Perejon, A; Raj, R; Pérez-Maqueda, LAJournal of the American Ceramic Society (2023) DOI: 10.1111/jace.19389
Abstract
This work presents an extension of the touch-free flash sintering technique. In the proposed technique, chemical reaction and sintering occur in a single step, without the use of electrodes, in the presence of electric and magnetic fields. We show that a dense, single-phase strontium hexaferrite magnet can be produced from a mixture of commercial carbonate and oxide powders in a single step in a little more than a minute. This new technique implies significant reduction in energy and time consumption (primarily because of ultrafast processing) relative to conventional sintering.
Agosto, 2023 · DOI: 10.1111/jace.19389
Reactividad de Sólidos
Seville history insight through their construction mortars
Perez-Rodriguez, JL; Perez-Maqueda, LA; Franquelo, ML; Duran, AJournal of Thermal Analysis and Calorimetry, (2023) DOI: 10.1007/s10973-023-12313-y
Abstract
Seville is intimately linked to its historic role and extensive cultural heritage. The city has been occupied by Romans, Arabs and Christians, who built important historical buildings. Roman (first-second centuries) and Arabic (eleventh century) buildings, medieval Shipyard (thirteenth century), San Isidoro and Santa Maria de las Cuevas monasteries (fifteenth century), Santa Maria de las Cuevas (fifteenth century modified in eighteenth century), El Salvador Church (eighteenth century), the Royal Ordnance building (eighteenth century) and Santa Angela de la Cruz convent (twentieth century) performed with lining mortars, and mortars used in building stones (City Hall and Marchena Gate), all of them located in Seville (Spain), have been studied. Ninety-four mortar samples (employed as structural, plaster, coating) originally used or applied in restoration processes have been collected to perform an archaeometry study. The ratio of CO2 mass loss to hydraulic water (H2O) mass loss, and the mineralogical characterization by X-ray diffraction has been used to compare the mortars used in the different historical periods. Mainly hydraulic mortars were widely used in all these studied monuments as most mortars showed CO2/H2O ratios within the 4-10 range. Moreover, the thermal analysis curves also showed a broad temperature range for the thermal decomposition of the carbonate fraction of the mortars.
Julio, 2023 · DOI: 10.1007/s10973-023-12313-y
Química de Superficies y Catálisis
New 3D Printing Strategy for Structured Carbon Devices Fabrication
Delgado-Martin, G; Rodriguez, N; Dominguez, MI; Agamez-Pertuz, YY; Tejada, MM; Ruiz-Lopez, E; Ivanova, S; Centeno, MACatalysts, 13 (2023) 1039 DOI: 10.3390/catal13071039
Abstract
This work shows a new method for the preparation of 100% carbon-structured devices. The method is based on resorcinol-formaldehyde polymerization, using starch as a binder with the addition of a certain amount of external carbon source before polymerization. Molds obtained by 3D printing are used to shape the structured devices in the desired shape, and the ultimate pyrolysis step consolidates and produces the carbonaceous devices. The proposed method allows obtaining supports with different textural and surface properties varying the carbonaceous source, the solvent, or the pyrolysis conditions, among other factors. The as-obtained devices have demonstrated their usefulness as palladium supports for the gas-phase formic acid dehydrogenation reaction. The monolith shows a high conversion of formic acid (81% according to H-2 production) and a high selectivity towards hydrogen production at mild temperatures (80% at 423 K).
Julio, 2023 · DOI: 10.3390/catal13071039
Reactividad de Sólidos
Thermochemical energy storage using calcium magnesium acetates under low CO2 pressure conditions
Amghar, N; Jimenez, PES; Maqueda, LAP; Perejon, AJournal of Energy Storage, 63 (2023) 106958 DOI: 10.1016/j.est.2023.106958
Abstract
The calcium looping multicycle performance of CaO-based materials, derived from calcium magnesium acetates with different Mg content were tested under experimental conditions compatible with thermochemical energy storage. In order to reduce the sintering-induced decay in performance, calcination at an absolute CO2 pressure of 0.1 bar and 0.01 bar is implemented. CaO carbonation is performed at standard 1 bar CO2 conditions. The samples can be fully calcined in short residence times. Samples with MgO present high cycling stability, even when the MgO content is as low as 5 mol%. The effective conversion values lie within the range 0.88-0.84 over ten calcination/carbonation cycles, which provides an accumulated energy storage density of 90.9 GJ/m3. This outstanding reactivity is related with the microstructure of the sample after calcination composed of CaO nanoparticles that are highly reactive for carbonation.
Julio, 2023 · DOI: 10.1016/j.est.2023.106958
Nanotecnología en Superficies y Plasma
Setting a comprehensive strategy to face the runback icing phenomena
Mora, J et al.Surface & Coatings Technology, 465 (2023) 129585 DOI: 10.1016/j.surfcoat.2023.129585
Abstract
The development of anti-icing robust surfaces is a hot topic nowadays and particularly crucial in the aeronautics or wind energy sectors as ice accretion can compromise safety and power generation efficiency. However, the current performance of most anti-icing strategies has been proven insufficient for such demanding applications, particularly in large unprotected zones, which located downstream from thermally protected areas, may undergo secondary icing. Herein, a new testing methodology is proposed to evaluate accretion mechanisms and secondary icing phenomena through, respectively, direct impact and running-wet processes and systematically applied to anti-icing materials including commercial solutions and the latest trends in the state-of-the-art. Five categories of materials (hard, elastomeric, polymeric matrix, SLIPS and superhydrophobic) with up to fifteen formulations have been tested. This Round-Robin approach provides a deeper understanding of anti-icing mechanisms revealing the strengths and weaknesses of each material. The conclusion is that there is no single passive solution for anti-ice protection. Thus, to effectively protect a given real component, different tailored materials fitted for each particular zone of the system are required. For this selection, shape analysis of such a component and the impact characteristics of water droplets under real conditions are needed as schematically illustrated for aeronautic turbines.
Julio, 2023 · DOI: 10.1016/j.surfcoat.2023.129585
Materiales Ópticos Multifuncionales
Ultrapure Green High Photoluminescence Quantum Yield from FAPbBr3 Nanocrystals Embedded in Transparent Porous Films
Romero-Pérez, C; Delgado, NF; Herrera-Collado, M; Calvo, ME; Míguez, H (MigChemistry of Materials, 35 (2023) 5541-5549 DOI: 10.1021/acs.chemmater.3c00934
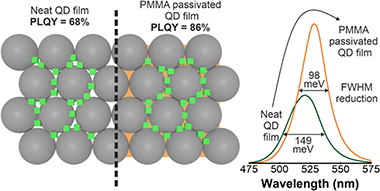
Abstract
Achieving highly transparent and emissive films based on perovskite quantum dots (PQDs) is a challenging task since their photoluminescence quantum yield (PLQY) typically drops abruptly when they are used as building blocks to make a solid. In this work, we obtain highly transparent films containing FAPbBr(3) quantum dots that display a narrow green emission (lambda = 530 nm, full width at half-maximum (FWHM) = 23 nm) with a PLQY as high as 86%. The method employed makes use of porous matrices that act as arrays of nanoreactors to synthesize the targeted quantum dots within their void space, providing both a means to keep them dispersed and a protective environment. Further infiltration with poly(methyl methacrylate) (PMMA) increases the mechanical and chemical stability of the ensemble and serves to passivate surface defects, boosting the emission of the embedded PQD and significantly reducing the width of the emission peak, which fulfills the requirements established by the Commission Internationale de l'E ' clairage (CIE) to be considered an ultrapure green emitter. The versatility of this approach is demonstrated by fabricating a color-converting layer that can be easily transferred onto a light-emitting device surface to modify the spectral properties of the outgoing radiation.
Julio, 2023 · DOI: 10.1021/acs.chemmater.3c00934
Química de Superficies y Catálisis
Effect of noble metal addition over active Ru/TiO2 catalyst for CO selective methanation from H2 rich- streams
Bobadilla, LF; Muñoz-Murillo, A; Gandara-Loe, J; Perez, A; Laguna, OH; Martinez, TLM; Penkova, A; Centeno, MA; Odriozola, JAInternational Journal of Hydrogen Energy, 48 (2023) 25065-25074 DOI: 10.1016/j.ijhydene.2022.07.072
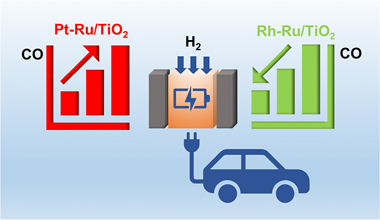
Abstract
Selective CO methanation from H2-rich stream has been regarded as a promising route for deep removal of low CO concentration and catalytic hydrogen purification processes. This work is focused on the development of more efficient catalysts applied in practical conditions. For this purpose, we prepared a series of catalysts based on Ru supported over titania and promoted with small amounts of Rh and Pt. Characterization details revealed that Rh and Pt modify the electronic properties of Ru. The results of catalytic activity showed that Pt has a negative effect since it promotes the reverse water gas shift reaction decreasing the selectivity of methanation but Rh increases remarkably the activity and selectivity of CO methanation. The obtained results suggest that RuRh-based catalyst could become important for the treatment of industrial-volume streams.
Julio, 2023 · DOI: 10.1016/j.ijhydene.2022.07.072
Reactividad de Sólidos
R-curve evaluation of 3YTZP/graphene composites by indirect compliance method
Lopez-Pernia, C; Munoz-Ferreiro, C; Prada-Rodrigo, J; Moreno, P; Reveron, H; Chevalier, J; Morales-Rodriguez, A; Poyato, R; Gallardo-Lopez, AJournal of the European Ceramic Society, 43 (2022) 3486-3497 DOI: 10.1016/j.jeurceramsoc.2023.02.002
Abstract
This work addresses the crack growth resistance of 3 mol% Yttria-doped Tetragonal Zirconia Polycrystalline (3YTZP) spark-plasma sintered (SPS) composites containing two types of graphene-based nanomaterials (GBN): exfoliated graphene nanoplatelets (e-GNP) and reduced graphene oxide (rGO). The crack growth resistance of the composites is assessed by means of their R-Curve behavior determined by three-point bending tests on single edge "V" notched beams (SEVNB), in two different orientations of the samples: with the crack path perpendicular or parallel to the pressure axis during the SPS sintering. The sharp edge notches were machined by ultrashort laser pulsed ablation (UPLA). The compliance and optical-based methods for evaluating the crack length are compared on the basis of the experimental R-Curve results in composites with 2.5 vol% rGO tested in the perpendicular orientation. Moreover, the activation of reinforcement mechanisms is evaluated by both the fracture surface inspection by Scanning Electron Microscopy and a compliance analysis. It is shown that the indirect compliance method is relevant and reliable for calculating the R-Curve of 3YTZP/GBN composites. The effect of the type and content of GBN on the crack growth resistance of the composites is also discussed.
Julio, 2023 · DOI: 10.1016/j.jeurceramsoc.2023.02.002
Nanotecnología en Superficies y Plasma
Dielectric multilayers for broadband optical rotation enhancement
Pellegrini, G; Mogni, E; Gil-Rostra, J; Yubero, F; Fossati, S; Dostálek, J; Vázquez, RM; Osellame, R; Celebrano, M; Finazzi, M; Biagioni, PNuovo Cimento C-Colloquia and Communications in Physics, 46 (2023) 111 DOI: 10.1393/ncc/i2023-23111-1
Abstract
We design a simple dielectric multilayer capable of sustaining broadband superchiral surface waves. We show that the platform can produce large optical chirality enhancements in a wavelength range of hundreds of nanometers. We finally demonstrate that these properties result in the enhancement of the optical rotation signal well above two orders of magnitude, thus extending surface-enhanced chiral spectroscopies beyond the traditionally addressed circular dichroism signals.
Julio, 2023 · DOI: 10.1393/ncc/i2023-23111-1
Fotocatálisis Heterogénea: Aplicaciones
ZnO/TiO2 and ZnO/Nb2O5 as effective systems for the treatment of enteric bacteria and commercial dyes
Hernandez, JS; Murcia, JJ; Rojas, H; Hidalgo, MC; Navio, JARevista Facultad de Ingeniería-Universidad de Antioquia, 108 (2023) 9-17 DOI: 10.17533/udea.redin.20220785
Abstract
In this study, ZnO/TiO2 and ZnO/Nb2O5 photocatalysts were evaluated in the river pollution remediation and wastewater treatment from textile factories, thus, the target pollutants selected for this study were enteropathogenic bacteria and commercial dyes. The mixed oxide systems were extensively analyzed in order to explore their physicochemical properties. From this analysis, it was found that the coupling of two oxides did not modify the crystallinity of the pristine semiconductors. As a result, XRD Wurtzite phase, hexagonal phase, and anatase phases were identified for ZnO, Nb2O5, and TiO2 photocatalyst, respectively. Using UV-Vis DRS, a higher absorption for mixed oxides in the visible region of the electromagnetic spectrum was observed, along with a decrease in the band gap value in these materials. The results of the photocatalytic activity evaluation showed that the coupling of ZnO with Nb2O5 and TiO2 increased the effectiveness of the total organic carbon (TOC) and E. Coli elimination. 83% of TOC and elimination of 64% of E. coli were achieved using ZnO/Nb2O5 photocatalyst for the treatment of water samples from the polluted river.
Julio, 2023 · DOI: 10.17533/udea.redin.20220785
Reactividad de Sólidos
An overview of polymeric composite scaffolds with piezoelectric properties for improved bone regeneration
Donate, R; Paz, R; Moriche, R; Sayagués, MJ; Alemán-Domínguez, ME; Monzón, MMaterials & Design, 231 (2023) 112085 DOI: 10.1016/j.matdes.2023.112085
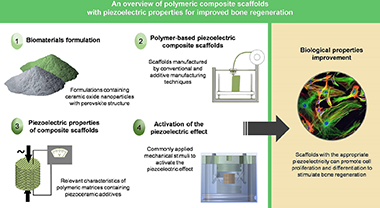
Abstract
Despite the dramatic change that Tissue Engineering or stem cell therapies have brought to current therapeutic strategies, there is a lack of functionalities in the available biomaterials for manufacturing scaffolds to treat several highly prevalent osseous diseases (osteochondral defects, osteoporosis, etc.). One promising approach to fill this gap involves the development of innovative piezoelectric scaffolds for improved bone regeneration. Scaffolds with the appropriate piezoelectricity can positively influence the proliferation and differentiation of mesenchymal stem cells to regenerate bone tissue, since surface electrical charges play a key role in the mechanotransduction process. In this work, polymeric-based composite scaffolds with piezoelectric properties intended for bone tissue engineering are reviewed. Special attention is paid to biocompatible, piezoelectric polymers that show suitable properties to be pro-cessed by additive manufacturing techniques. Previous works on composite scaffolds based of these poly-meric matrices and containing piezoceramic additives are summarized. The use of piezoelectric nanostructured composite formulations containing lead-free ceramic oxide nanoparticles with per-ovskite structure is highlighted. Also, different commonly applied mechanical stimuli to activate the piezoelectric effect of the developed materials are presented. Finally, other applications of such scaffolds are mentioned, including their capabilities for real-time monitoring
Julio, 2023 · DOI: 10.1016/j.matdes.2023.112085
Química de Superficies y Catálisis
Biochar production from cellulose under reductant atmosphere: influence of the total pyrolysis time
Santos, JL; Centeno, MA; Odriozola, JARSC Advances, 13 (2023) 21071-21079 DOI: 10.1039/d3ra03093h
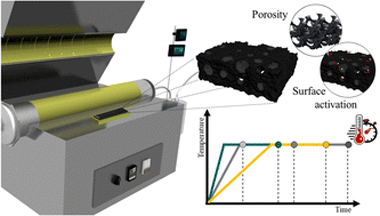
Abstract
Today's rising energy costs, coupled with increasing energy demand, make it necessary to search for more efficient energy processes. In recent years, there have been increasing efforts to develop efficient catalysts based on waste-derived char, by a single step where the carbon precursor and the metallic active phase one undergo a single common thermal process under a reductant atmosphere at high temperature. The use of a reductant atmosphere drives the formation of carbonaceous materials with different characteristics than those obtained under the standard nitrogen-inert one. Our work evaluates the influence of the residence time and the heating rate on the physicochemical properties of the biochar obtained. Relatively long residence times and slow heating rates, improve the yield to the resulting biochar, without increasing production cost, making the subsequent char-based metallic catalyst synthesis more efficient. The heating rate was shown to be key in improving the properties of the char in a smoother and more controlled way, unlocking a new working pathway for the efficient design and production of char-based catalysts in a one-pot synthesis.
Julio, 2023 · DOI: 10.1039/d3ra03093h
Materiales Avanzados
Influence of firing temperature on the ceramic properties of illite-chlorite-calcitic clays
Martinez-Martinez, S; Perez-Vilarejo, L; Garzon, E; Sanchez-Soto, PJCeramics International, 49 (2023) 24541-24557 DOI: 10.1016/j.ceramint.2022.11.077
Abstract
The influence of firing temperature on the ceramic properties of illite-chlorite-calcitic clays has been investigated. Three samples of the same clay deposit have been selected. Weight loss, dimensional changes, water absorption, bulk density, open porosity, flexural and compressive strengths, initial capillary water absorption rate and thermal conductivity have been determined as a function of firing temperature in the range 900-1200 degrees C with 1 h of soaking time. The microstructures of the fired samples have been examined by SEM and the phase evolution studied by XRD. The water absorption capacity decreased from similar to 22% at 900 degrees C/1 h to a maximum of 12% at 1200 degrees C/1 h with a maximum linear shrinkage of similar to 2.7%. The open porosities decreased from similar to 22% at 900 degrees C/1 h up to similar to 20% at 1200 degrees C/1 h as an effect of progressive sintering with higher densification degree of the ceramic bodies. The flexural strength reached a maximum value of similar to 34 MPa at 1200 degrees C/1 h. In contrast, the compressive strengths increased by firing up to a maximum of similar to 114 MPa at 1200 degrees C/1 h. The thermal conductivity increased slightly as increasing firing temperature with a maximum value of 0.582 W/m.K in samples fired at 1200 degrees C/1 h. The Ryshkevitch-Duckworth equation was applied and the results indicated that compressive strength is related linearly with open porosity. A linear correlation was found between thermal conductivity and open porosity. The microstructural evolution by SEM indicated that there is a change of the fired samples at 1100 degrees C as compared to SEM observations at 900 and 1000 degrees C. There is an increase of contacts between particles and layered structures associated to dehydroxylated clay minerals (illite and chlorite), quartz particles and pores developed by firing. At 1200 degrees C/1 h, the microstructures have changed associated to the higher degree of vitrification in the fired sample, with consolidation of the material, interparticle and neck contacts with formation of vitrified bridges. The formation of closed and large open pores of several sizes has been achieved by firing. Small particles were observed as a fine precipitation of crystals in the vitrified structures associated to anorthite, hematite and quartz relicts. This change in microstructure allowed deduce that the compressive strength increased upon firing, with maximum values of this ceramic property at 1200 degrees C. The ceramic bodies were more sintered by firing and the open porosity decreased progressively. Brickmaking is the main application of these fired illite-chlorite calcitic clays. These clays fired at 900-1100 degrees C, with 1 h of soaking time, could be applied in the fabrication of clay roofing tiles, tiles and even porous ceramic supports with small variations on shrinkage and porosity, good flexural strengths and high compressive strengths. Samples fired at higher temperatures, 1100 degrees C/1 h, could be applied as ceramic bricks showing a medium porosity (similar to 20%). They show almost the same bulk density when they are fired at lower temperatures (900 degrees C). Samples fired at higher temperatures (1150-1200 degrees C/1 h) could be applied as dark ceramic products. This investigation was interesting because a better knowledge of illite-chlorite-calcitic clays applied as ceramic raw materials has been achieved.
Julio, 2023 · DOI: 10.1016/j.ceramint.2022.11.077
Reactividad de Sólidos
Partial oxycombustion-calcium looping hybridisation for CO2 capture in waste-to-energy power plants
Ortiz, C; García-Luna, S; Chacartegui, R; Valverde, JM; Pérez-Maqueda, LJournal of Cleaner Production, 403 (2023) 136776 DOI: 10.1016/j.jclepro.2023.136776
Abstract
Integrating bioenergy and carbon capture and storage (BECCS) presents a great opportunity for power produc-tion with negative global CO2 emissions. This work explores a novel synergetic system that integrates mem-branes, partial biomass oxycombustion and the calcium looping (CaL) process. Polymeric membranes generate oxygen-enriched air (OEA) with an O2 concentration of 39%v/v, which is used for partial oxycombustion of biomass waste. The CO2-enriched flue gas evolves from the waste-to-energy plant to the CaL unit, where CO2 concentration is increased up to 90-95%v/v, ready for purification and sequestration. Compared to only oxy-combustion systems, the proposed concept presents fewer technological challenges in retrofitting boilers to waste-to-energy plants. Moreover, this new approach is highly efficient as integrating membranes to produce OEA instead of cryogenic distillation systems significantly reduces energy consumption. A novel integration concept is modelled to evaluate the whole process efficiency and the effect of key parameters on the system performance, such as the temperature of the reactors, the membrane surface area, and the partial oxy-combustion degree. The results show that the so-called mOxy-CaL system has an energy consumption associ-ated with CO2 capture below 4 MJ/kg CO2 (a 31% lower than that for a conventional CaL process), with a higher CO2 capture efficiency than oxycombustion and the CaL process separately. On the other hand, the economic analysis shows a higher CO2 capture cost for the novel configuration than for the typical CaL configuration due to the additional investment cost of the membrane system. Improvements in membrane performance by increasing its permeance and diminishing the required surface area would significantly reduce the economic cost of this novel integration. Using membranes with permeance over 400 GPU would boost the system's competitiveness.
Junio, 2023 · DOI: 10.1016/j.jclepro.2023.136776
Química de Superficies y Catálisis
Hydrothermal carbonization vs. anaerobic digestion to valorize fruit and vegetable waste: A comparative technical and energy assessment
Metyouy, K; Gonzalez, R; Gomez, X; Gonzalez-Arias, J; Martinez, EJ; Chafik, T; Sanchez, ME; Cara-Jiménez, JJournal of Environmental Chemical Engineering, 11 (2023) 109925 DOI: 10.1016/j.jece.2023.109925
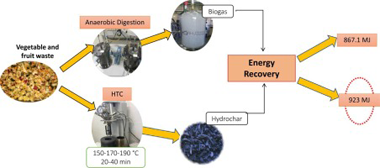
Abstract
Herein, the valorization of vegetable and fruit waste was assessed via hydrothermal carbonization (HTC) and anaerobic digestion (AD) in terms of product characterization and energy requirements. HTC was conducted at reaction temperatures between 150 & DEG;C and 190 & DEG;C, and residence times between 20 min and 40 min. The increase in the process severity resulted in hydrochars with higher carbon contents and higher energy densification ratios. AD was performed in two different ways. i.e., batch and semi-continuous reactions. From the batch experiments a methane yield of 300 L CH4/kg VS was obtained, while for the semi-continuous, the average specific methane production estimated (for HRTs from 75 to 50 days) was 213 & PLUSMN; 32 L CH4/kg VS. To estimate the energy re-quirements, mass and energy balances were performed considering the basic stages of each process to obtain a suitable biofuel material. In this sense, it was concluded that for this specific waste, AD was a more suitable process with a positive energy net balance. On the contrary, HTC presented a negative energy net balance being required 1.29 MJ/kg of fresh food waste. A combined HTC-AD treatment may be an efficient method to take advantage of both technologies leading to higher energy efficiencies and other valuable products.
Junio, 2023 · DOI: 10.1016/j.jece.2023.109925
Química de Superficies y Catálisis
Are Ni/ and Ni5Fe1/biochar catalysts suitable for synthetic natural gas production? A comparison with g-Al2O3 supported catalysts
González-Castaño, M; Morales, C; de Miguel, JCN; Boelte, JH; Klepel, O; Flege, JI; Arellano-Garcia, HGreen Energy & Environment, 8 (2023) 744-756 DOI: 10.1016/j.gee.2021.05.007
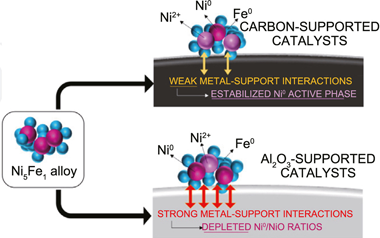
Abstract
Among challenges implicit in the transition to the post-fossil fuel energetic model, the finite amount of resources available for the technological implementation of CO2 revalorizing processes arises as a central issue. The development of fully renewable catalytic systems with easier metal recovery strategies would promote the viability and sustainability of synthetic natural gas production circular routes. Taking Ni and NiFe catalysts supported over g-Al2O3 oxide as reference materials, this work evaluates the potentiality of Ni and NiFe supported biochar catalysts for CO2 methanation. The development of competitive biochar catalysts was found dependent on the creation of basic sites on the catalyst surface. Displaying lower Turn Over Frequencies than Ni/Al catalyst, the absence of basic sites achieved over Ni/C catalyst was related to the depleted catalyst performances. For NiFe catalysts, analogous Ni5Fe1 alloys were constituted over both alumina and biochar supports. The highest specific activity of the catalyst series, exhibited by the NiFe/C catalyst, was related to the development of surface basic sites along with weaker NiFe-C interactions, which resulted in increased Ni0:NiO surface populations under reaction conditions. In summary, the present work establishes biochar supports as a competitive material to consider within the future low-carbon energetic panorama.
Junio, 2023 · DOI: 10.1016/j.gee.2021.05.007
Química de Superficies y Catálisis
Review and Perspectives of CO2 Absorption by Water- and Amine-Based Nanofluids
Yuan, CT; Wang, Y; Baena-Moreno, FM; Pan, Z; Zhang, R; Zhou, H; Zhang, ZEnergy & Fuels, 37 (2023) 8883-8901 DOI: 10.1021/acs.energyfuels.3c00874
Abstract
The emission of greenhouse gases, especially CO2, hasbecome a major cause of environmental degradation, and carbon capture,utilization, and storage (CCUS) is a proposed solution to mitigateits impact. Nanofluids, a relatively new method for CO2 absorption, have gained attention in recent years. This review focuseson conventional methods for preparing nanofluids along with techniquesto improve their stability and enhance the CO2 absorptionand desorption mechanisms. Additionally, the influences of factors,i.e., nanoparticle and base solution types as well as nanoparticleconcentration, on the CO2 absorption process are summarized.Furthermore, models that can predict the absorption of CO2 accurately are outlined. It is found that the types of both baseliquids and nanoparticles have an important impact on the absorptionby nanofluids. In-depth studies on the predictive capabilities ofartificial intelligence (AI) models hold immense potential in thisregard. This review also puts forth effective strategies to addressprevailing challenges. This will provide a solid theoretical basisfor this field and underscore the promising potential of nanofluidsas CO2 solvents. There are still many unexplored aspectsto be considered, such as the economic viability and energy consumptionof this technology.
Junio, 2023 · DOI: 10.1021/acs.energyfuels.3c00874
Química de Superficies y Catálisis
Toluene combustion on MnOx, CeO2, and Mn-Ce-O solids prepared via citrate complexation, and citrate and urea combustion methods
Rahou, S; Benadda-Kordjani, A; Ivanova, S; Odriozola, JA; Chebout, R; Mahzoul, H; Zouaoui, NJournal of Nanoparticle Research, 25 (2023) 114 DOI: 10.1007/s11051-023-05759-6
Abstract
MnOx, CeO2, and MnCe-O (Mn/Ce = 1) solids have been prepared via the citrate complexation and combustion method using citrate and urea precursors. The solids have been characterized by XRD, SEM-EDX, N-2-adsorption-desorption, UV-Vis spectroscopy, TPR, O-2-TPD, and XPS techniques. The catalytic reactivity of the manganese oxides was not affected by the preparation protocol. In the case of ceria and mixed oxides, the synthesis method greatly affected the structural and chemical properties, ultimately altering their reactivity. The citrate complexation method produced the most homogeneous and active mixed oxide, whereas the urea combustion method resulted in less active solids. The mixed oxide prepared via urea combustion was less active than the manganese single oxide; the decrease in activity was attributed to phase separation and the formation of Mn3O4 domains on the surface of ceria. In contrast, citrate complexation resulted in solids with the lowest particle size (similar to 3 nm), the highest oxidation state for manganese, and the highest proportion of oxygen vacancies, which promote the oxidation reaction.
Junio, 2023 · DOI: 10.1007/s11051-023-05759-6
Química de Superficies y Catálisis
Formic Acid Dehydrogenation over a Monometallic Pd and Bimetallic Pd:Co Catalyst Supported on Activated Carbon
Pelaez, MR; Ruiz-Lopez, E; Dominguez, MI; Ivanova, S; Centeno, MACatalysts, 13 (2023) 977 DOI: 10.3390/catal13060977
Abstract
In this study, palladium is proposed as an active site for formic acid dehydrogenation reaction. Pd activity was modulated with Co metal with the final aim of finding a synergistic effect that makes possible efficient hydrogen production for a low noble metal content. For the monometallic catalysts, the metal loadings were optimized, and the increase in the reaction temperature and presence of additives were carefully considered. The present study aimed, to a great extent, to enlighten the possible routes for decreasing noble metal loading in view of the better sustainability of hydrogen production from liquid organic carrier molecules, such as formic acid.
Junio, 2023 · DOI: 10.3390/catal13060977
Nanotecnología en Superficies y Plasma
Structure and Void Connectivity in Nanocolumnar Thin Films Grown by Magnetron Sputtering at Oblique Angles
Alvarez, R; Regodon, G; Acosta-Rivera, H; Rico, V; Alcala, G; Gonzalez-Elipe, AR; Palmero, ACoatings, 13 (2023) 991 DOI: 10.3390/coatings13060991
Abstract
The morphology and void connectivity of thin films grown by a magnetron sputtering deposition technique at oblique geometries were studied in this paper. A well-tested thin film growth model was employed to assess the features of these layers along with experimental data taken from the literature. A strong variation in the film morphology and pore topology was found as a function of the growth conditions, which have been linked to the different collisional transport of sputtered species in the plasma gas. Four different characteristic film morphologies were identified, such as (i) highly dense and compact, (ii) compact with large, tilted mesopores, (iii) nanocolumns separated by large mesopores, and (iv) vertically aligned sponge-like coalescent nanostructures. Attending to the topology and connectivity of the voids in the film, the nanocolumnar morphology was shown to present a high pore volume and area connected with the outside by means of mesopores, with a diameter above 2 nm, while the sponge-like nanostructure presented a high pore volume and area, as well as a dense network connectivity by means of micropores, with a diameter below 2 nm. The obtained results describe the different features of the porous network in these films and explain the different performances as gas or liquid sensors in electrochromic applications or for infiltration with nanoparticles or large molecules.
Junio, 2023 · DOI: 10.3390/coatings13060991
Reactividad de Sólidos
Effect of thermal treatments below devitrification temperature on the magnetic and magnetocaloric properties in mechanically alloyed Fe70Zr30 powders
Manchon-Gordon, AF; Blazquez, JS; Kowalczyk, M; Ipus, JJ; Kulik, T; Conde, CFJournal of Non-Crystalline Solids, 609 (2023) 122267 DOI: 10.1016/j.jnoncrysol.2023.122267
Abstract
In this work, the relaxation of the amorphous structure of mechanically alloyed Fe70Zr30 powders has been analyzed through interrupted heating ramps below the devitrification temperature. As a result of such thermal treatment, Curie temperature and temperature at maximum magnetic entropy change curves shift to higher temperatures as the temperature of heating treatment increases. This effect can be attributed to both the release of the stress accumulated in the amorphous powder during the milling process and to the initiation of nucleation of alpha-Fe crystallites, as it has been shown by Mo center dot ssbauer spectroscopy.
Junio, 2023 · DOI: 10.1016/j.jnoncrysol.2023.122267
Nanotecnología en Superficies y Plasma - Tribología y Protección de Superficies
Low temperature nucleation of thermochromic VO2 crystal domains in nanocolumnar porous thin films
Alcaide, AM; Regodon, G; Ferrer, FJ; Rico, V; Alvarez, R; Rojas, TC; Gonzalez-Elipe, AR; Palmero, ANanotechnology, 34 (2023) 255702 DOI: 10.1088/1361-6528/acc664
Abstract
The low temperature formation of monoclinic VO2 crystal domains in nanocolumnar vanadium/oxygen thin films prepared by magnetron sputtering at oblique angles is analyzed. The synthesis procedure involved the deposition of amorphous nanocolumnar VO1.9 thin films at room temperature and its subsequent annealing at temperatures between 250 °C and 330 °C in an oxygen atmosphere. The thermochromic transition of these films was found at a temperature of 47 °C when the annealing temperature was 270 °C and 58 °C when the annealing temperature was 280 °C and 290 °C, presenting a clear drop of the optical transmittance in the infrared region of the spectrum. The significant downshift in the temperature window to obtain VO2 in comparison with compact films and other strategies in literature is explained by the particular morphology of the nanocolumnar structures, which contains numerous defects along with open and embedded porosity.
Junio, 2023 · DOI: 10.1088/1361-6528/acc664
Nanotecnología en Superficies y Plasma - Materiales Nanoestructurados y Microestructura
Microstructural characterization and thermal stability of He charged amorphous silicon films prepared by magnetron sputtering in helium
Fernández, A; Sauvage, T; Diallo, B; Hufschmidt, D; de Haro, MCJ; Montes, O; Martínez-Blanes, JM; Caballero, J; Godinho, V; Ferrer, FJ; Ibrahim, S; Brault, P; Thomann, ALMaterials Chemistry and Physics, 301 (2023) 127674 DOI: 10.1016/j.matchemphys.2023.127674
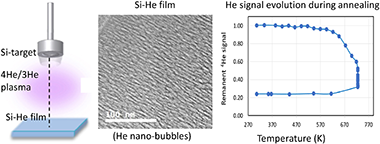
Abstract
Sputtering of silicon in a Helium magnetron discharge has been reported as a bottom-up procedure to obtain amorphous Si films containing high amounts of gas-filled nanopores. Here we compare the microstructure and composition of Si-He nanocomposite films deposited by magnetron sputtering (MS) with 4He in DC or RF and 3He in RF operation modes. Electron microscopy (SEM and TEM), X-ray diffraction (XRD) and ion beam analysis (IBA) have been used to analyze the films and to investigate the in-situ and ex-situ thermal evolution. Depending on deposition conditions different in depth compositions, nanopore size and shape distributions, porosity and He content could be obtained. The presence of impurities (i.e. oxygen) has shown to promote He diffusivity reducing He accumulation. The start temperature of He-release varied in the range 473-723 K without films crystallization. Films grown in RF mode reached contents of 32 and 29 at% of 4He and 3He and were respectively stable up to 573 and 723 K both in vacuum and under inert gas flow. In-situ p-EBS (proton Elastic Back Scattering) allowed monitoring the He release accompanied by blistering/delamination effects visualized by SEM. These results show the potentiality of annealing to hold nano-porous structures after liberation of trapped gas.
Junio, 2023 · DOI: 10.1016/j.matchemphys.2023.127674
Reactividad de Sólidos
A practical analysis for decelerated growth processes to get physically meaningful kinetic parameters from classical nucleation and growth theory despite of overgrowth
Blazquez, JS; Caballero-Flores, R; Manchon-Gordon, AF; Borrego, JM; Conde, CFJournal of Non-Crystalline Solids, 610 (2023) 122305 DOI: 10.1016/j.jnoncrysol.2023.122305
Abstract
We have analyzed the overgrowth problem arising in decelerated growth processes of spherical crystals in the frame of classical nucleation and growth theory developed by Kolmogorov, Johnson and Mehl, and Avrami (KJMA). To do that, simulations of decelerated growth transformations with a constant nucleation rate have been performed, changing the linear growth rate of spherically shaped nuclei from null (instantaneous growth rate) to constant (characteristic of interface controlled growth processes). We propose the determination of the actual kinetic parameters through the analysis of the inflection point of time evolution of transformed fraction. The correlations found between the effective kinetic parameters from direct KJMA analysis and the actual ones make it possible obtaining physically meaningful parameters. The proposed analysis has been applied to the nano-crystallization of amorphous FINEMET-type compositions.
Junio, 2023 · DOI: 10.1016/j.jnoncrysol.2023.122305
Reactividad de Sólidos
Reversibility and thermal dependence of the martensitic transformation in a melt-spun Ni55Fe17Ga26Co2 Heusler alloy
Manchon-Gordon, AF; Vidal-Crespo, A; Blazquez, JS; Kowalczyk, M; Ipus, JJ; Kulik, T; Conde, CFJournal of Alloys and Compounds, 946 (2023) 169484 DOI: 10.1016/j.jallcom.2023.169484
Abstract
An almost single phase 14 M modulated martensite is obtained in melt spun ribbon of Ni55Fe17Ga26Co2 Heusler alloy. The effect of thermal treatments on the stability of the reverse martensitic transformation from 14 M modulated martensite to austenite phase in this system has been investigated by both non -isothermal and isothermal treatments. Heating above martensitic transformation promotes a continuous reduction of the martensitic transformation temperature, which stabilizes the austenite phase at room temperature and induces the precipitation of the gamma phase. However, thermal treatments at tem-peratures between the austenite start and finish temperatures induce the decoupling of the austenite formation in a subsequent heating. The two successive reverse martensitic transformations could be as-cribed to the untransformed martensite in the previous interrupted heating and to the new martensite formed during cooling.
Junio, 2023 · DOI: 10.1016/j.jallcom.2023.169484
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Catalytic performance of cobalt supported onto APTES functionalized TiO2 for Fischer-Tropsch reaction
Platero, F; Caballero, A; Colon, GFuel, 340 (2023) 127528 DOI: 10.1016/j.fuel.2023.127528
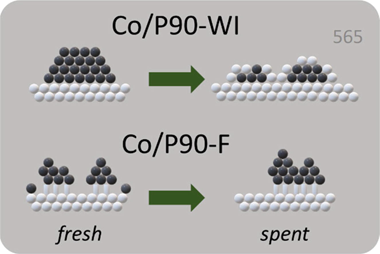
Abstract
Cobalt supported TiO2 catalysts have been prepared by wet-impregnation and by immobilization over APTES (3-aminopropyl triethoxysilane) grafted TiO2. Impregnated system showed better catalytic performance after reduction at 260 degrees C but significant deactivation is observed. On the contrary, functionalized catalyst showed better catalytic performance after reduction at 400 degrees C with notable stability. We have stated from CO-DRIFT operando analysis that impregnated system is strongly affected by negative SMSI (strong metal-support inter-action) upon reduction at higher temperature. While immobilization on APTES hinders the loss of metal active sites. The study of spent catalysts denotes that Co is redispersed in the impregnated catalyst while functionalized trends to form agglomerates.
Mayo, 2023 · DOI: 10.1016/j.fuel.2023.127528
Materiales Avanzados
Characterization, thermal and ceramic properties of clays from Alhabia (Almeria, Spain)
Rat, E; Martinez-Martinez, S; Sanchez-Garrido, JA; Perez-Vilargejo, L; Garzon, E; Sanchez-Soto, PJCeramics International, 49 (2023) 14814-14825 DOI: 10.1016/j.ceramint.2022.05.328
Abstract
Clays from Alhabia (Almeria, Spain) have been investigated in this work using several analytical techniques: X-Ray Diffraction (XRD), X-Ray Fluorescence (XRF), thermal analysis (Thermogravimetry, TG, and its first deriv-ative, DTG), Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray Spectroscopy (EDS). Texture characteristics (granulometry) and plasticity have been examined. The main ceramic properties (firing shrinkage, water absorption, bulk density, open porosity, flexural strength and thermal conductivity) have been determined using pressed and fired clay samples. Thus, the mineralogical, chemical, textural and ceramic features of these clays have been evidenced for the first time. The mineralogical analysis by XRD indicated that the clay samples are constituted by a mixture of chlorite and illite, as main clay minerals, being quartz and other minerals in lower relative proportion (calcite, gypsum and hematite). This finding is important because the investigations on chlorite-illite-calcitic clays are very scarce. The chemical analysis by XRF showed that silica and alumina are predominant, as expected by the mineralogy, with medium contents of calcium oxide, from calcite, and alkalis, from illite, being-8 and-5%, respectively, besides iron and titanium oxides (-8%). The particle size analysis showed 71.76% of "clay fraction" (<2 mu m) and 21.66% of silt fraction (2-50 mu m). The plasticity index (Atterberg) was 14.3%, with acceptable moulding and extrusion properties. Thermal analysis by TG/DTG indicated a weight loss associated to dehydroxylation of structural water of the clay minerals and decarbonation of calcite by progressive heating. After the characterization of raw clays, the next step was the determination of ceramic properties of mixed and ground clays after firing using pressed bodies. For this purpose, two firing temperatures were selected (900 and 1100 degrees C) for 1 h. The examination of the resultant fired bodies indicated that porous ceramic materials (-36% open porosity and-22% of water absorption capacity) can be obtained by firing at 900 degrees C, with small variations in dimensions (<0.8% at 1100 degrees C). The porosity changed at relatively lower values by firing at 1100 degrees C (-34-35%), being associated to the presence of decomposed calcite. Bulk density was found almost constant from 900 to 1100 degrees C, with a maximum value of-1.67 g/cm3 at 1100 degrees C. Flexural strength reached a maximum value of 34.47 MPa at 1100 degrees C for the ground sample. Finally, thermal conductivity after firing the clay bodies was found almost constant at 900 and 1100 degrees C (0.457 and 0.479 W/mK, respectively). Taking into account these results, the main applications of the Alhabia clays have been evaluated. These clays can be used for the fabrication of porous ceramic supports and tiles by firing at 900 degrees C. Firing the clays at higher temperature (1100 degrees C) is of great interest for the fabrication of ceramic tiles and ceramic bricks of higher flexural strength with variable porosity and practically constant in dimensions. It is economically important although at higher processing costs. Finally, it can be emphasized that this work is a contribution of a better scientific knowledge of chlorite-illite-calcitic clays as ceramic raw materials.
Mayo, 2023 · DOI: 10.1016/j.ceramint.2022.05.328
Química de Superficies y Catálisis
MIL-100(Fe)-derived catalysts for CO2 conversion via low- and high-temperature reverse water-gas shift reaction
Loe, JG; Pena, AP; Espejo, JLM; Bobadilla, LF; Reina, TR; Pastor-Perez, LHeliyon, 9 (2023) e16070 DOI: 10.1016/j.heliyon.2023.e16070
Abstract
Fe-derived catalysts were synthesized by the pyrolysis of MIL-100 (Fe) metal-organic framework (MOF) and evaluated in the reverse water-gas shift (RWGS) reaction. The addition of Rh as a dopant by in-situ incorporation during the synthesis and wet impregnation was also considered. Our characterization data showed that the main active phase was a mixture of & alpha;-Fe, Fe3C, and Fe3O4 in all the catalysts evaluated. Additionally, small Rh loading leads to a decrease in the particle size in the active phase. Despite all three catalysts showing commendable CO selectivity levels, the C@Fe* catalyst showed the most promising performance at a temperature below 500 degrees C, attributed to the in-situ incorporation of Rh during the synthesis. Overall, this work showcases a strategy for designing novel Fe MOF-derived catalysts for RWGS reaction, opening new research opportunities for CO2 utilization schemes.
Mayo, 2023 · DOI: 10.1016/j.heliyon.2023.e16070
Química de Superficies y Catálisis
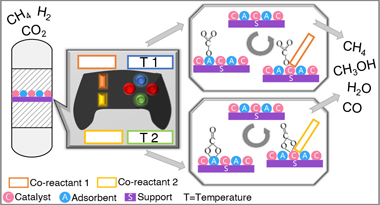
Unravelling the CO2 capture and conversion mechanism of a NiRu-Na2O switchable dual-function material in various CO2 utilisation reactions
Merkouri, LP; Martin-Espejo, JL; Bobadilla, LF; Odriozola, JA; Penkova, A; Reina, T; Duyar, MSJournal of Materials Chemistry A, 11 (2023) 13209-13216 DOI: 10.1039/d3ta01892j
Abstract
Time-resolved operando DRIFTS-MS was performed to elucidate the CO2 capture and conversion mechanisms of a NiRuNa/CeAl DFM in CO2 methanation, reverse water-gas shift, and dry reforming of methane. CO2 was captured mainly in the form of carbonyls and bidentate carbonates, and a spillover mechanism occurred to obtain the desired products.
Mayo, 2023 · DOI: 10.1039/d3ta01892j
Materiales Ópticos Multifuncionales
Modeling Weakly Scattering Random Media: A Tool to Resolve the Internal Structure of Nanoporous Materials
Jimenez-Solano, A; Miranda-Munoz, JM; Carretero-Palacios, S; Miguez, HAdvanced Photonics Research, 4 (2023) 5 DOI: 10.1002/adpr.202200267
Abstract
Nanoporous media scatter a small fraction of the light propagating through them, even if pore sizes are significantly smaller than the characteristic visible wavelengths. The disordered spatial modulation of the refractive index at the few or few tens of nanometers length scale, resulting from the presence of randomly distributed air bubbles or solid aggregates within a continuous solid background, gives rise to these weak scattering effects. However, standard theoretical approaches to describe this kind of media use effective medium approximations that do not account for diffuse, ballistic, and specular components. Herein, all spectral components and the angular distribution of the scattered light are captured through optical modeling. A Monte Carlo approach, combining scattering Mie theory and Fresnel equations, implemented within a genetic algorithm, allows us to decode the void and aggregate size distribution and hence the internal structure of a nanocrystalline titania (TiO2) film chosen as a paradigmatic example. The approach allows to generically describe the scattering properties of nanoporous materials which, as shown herein, may be used to decipher their internal structure from the fitting of their far-optical field properties.
Mayo, 2023 · DOI: 10.1002/adpr.202200267
Nanotecnología en Superficies y Plasma
Optical monitoring of detergent pollutants in greywater
Lahoz, F; de Armas-Rillo, S; Hernandez-Rodriguez, C; Gil-Rostra, J; Yubero, FOptics Express, 31 (2023) 15227-15238 DOI: 10.1364/OE.466194
Abstract
Large amount of wastewater is produced by washing machines and dishwashers, which are used in a daily basis. This domestic wastewater generated in households or office buildings (also called greywater) is drained directly to the drainpipes without differentiation from that with fecal contamination from toilets. Detergents are arguably the pollutants most frequently found in greywater from home appliances. Their concentrations vary in the successive stages in a wash cycle, which could be taken into account in a rational design of home appliances wastewater management. Analytical chemistry procedures are commonly used to determine the pollutant content in wastewater. They require collecting samples and their transport to properly equipped laboratories, which hampers real time wastewater management. In this paper, optofluidic devices based on planar Fabry-Perot microresonators operating in transmission mode in the visible and near infrared spectral ranges have been studied to determine the concentration of five brands of soap dissolved in water. It is found that the spectral positions of the optical resonances redshift when the soap concentration increases in the corresponding solutions. Experimental calibration curves of the optofluidic device were used to determine the soap concentration of wastewater from the successive stages of a washing machine wash cycle either loaded with garments or unloaded. Interestingly, the analysis of the optical sensor indicated that the greywater from the last water discharge of the wash cycle could be reused for gardening or agriculture. The integration of this kind of microfluidic devices into the home appliances design could lead to reduce our hydric environmental impact.
Mayo, 2023 · DOI: 10.1364/OE.466194
Química de Superficies y Catálisis
Carbon Capture Enhancement by Water-Based Nanofluids in a Hollow Fiber Membrane Contactor
Yuan, CT; Pan, Z; Wang, Y; Baena-Moreno, FM; Constantinou, A; Zhang, ZEnergy Technology, 11 (2023) 2300254 DOI: 10.1002/ente.202300254
Abstract
Nanoparticles are being used in the CO2 solvents to improve the capture performance. Herein, a 2D model is proposed to study the CO2 capture performance from a gaseous mixture using a hollow fiber membrane contactor (HFMC). Both water-based nanofluids of carbon nanotubes (CNT) and SiO2 are deployed as the carbon absorbents. It is verified that Brownian motion and grazing effect are the major reasons to enhance the mass transfer of nanofluids. The simulation findings show that the modeling data conform well with the experimental studies. The root-mean-square errors for SiO2 nanofluid and CNT nanofluid are 2.37% and 2.56%, respectively. When the amounts of nanoparticles increase between 0.02 and 0.06 wt%, CO2 capture efficiencies of SiO2 and CNT nanofluids increase by 7.92% and 13.17%, respectively. Also, the CNT nanofluid has a better capture performance than the SiO2 nanofluid. Furthermore, research is conducted into how membrane characteristics affect HFMC performance. It is indicated that increasing the membrane porosity and decreasing the membrane tortuosity have a positive impact on the capture efficiency. This work demonstrates the potentials in the use of nanoparticles in CO2 solvents and provides a solid theoretical basis for nanofluids to significantly enhance gas absorption.
Mayo, 2023 · DOI: 10.1002/ente.202300254
Química de Superficies y Catálisis
The Need for Flexible Chemical Synthesis and How Dual-Function Materials Can Pave the Way
Merkouri, LP; Paksoy, AI; Reina, TR; Duyar, MSACS Catalysis, 13 (2023) 7230-7242 DOI: 10.1021/acscatal.3c00880
Abstract
Since climate change keeps escalating, it is imperativethat theincreasing CO2 emissions be combated. Over recent years,research efforts have been aiming for the design and optimizationof materials for CO2 capture and conversion to enable acircular economy. The uncertainties in the energy sector and the variationsin supply and demand place an additional burden on the commercializationand implementation of these carbon capture and utilization technologies.Therefore, the scientific community needs to think out of the boxif it is to find solutions to mitigate the effects of climate change.Flexible chemical synthesis can pave the way for tackling market uncertainties.The materials for flexible chemical synthesis function under a dynamicoperation, and thus, they need to be studied as such. Dual-functionmaterials are an emerging group of dynamic catalytic materials thatintegrate the CO2 capture and conversion steps. Hence,they can be used to allow some flexibility in the production of chemicalsas a response to the changing energy sector. This Perspective highlightsthe necessity of flexible chemical synthesis by focusing on understandingthe catalytic characteristics under a dynamic operation and by discussingthe requirements for the optimization of materials at the nanoscale.
Mayo, 2023 · DOI: 10.1021/acscatal.3c00880
Reactividad de Sólidos
Influence of AC fields and electrical conduction mechanisms on the flash-onset temperature: Electronic (BiFeO3) vs. ionic conductors (8YSZ)
Molina-Molina, S; Perejón, A; Pérez-Maqueda, LA; Sánchez-Jiménez, PECeramics International, 49 (2023) 14834-14843 DOI: 10.1016/j.ceramint.2022.06.242
Abstract
This work aims to clarify the influence of AC (up to 50 kHz) vs DC fields on the flash-onset temperature, emphasizing the role of the electrical conduction mechanism. BiFeO3 (BFO) is used as an example of electronic conductor while 8-mol % Yttria-stabilized zirconia (8YSZ) is used as an example of ionic conductor. For 8YSZ, a frequency dependence of the flash-onset temperature and flash-induced heating is observed. This is consistent with the different contributions found in the total electrical response of 8YSZ as characterized by impedance spectroscopy measurements. Estimations based on the blackbody radiation model suggest that 8YSZ samples attain higher temperatures under AC fields due to a more efficient heating. Moreover, a noticeable decrease in the activation energy for the electrical conduction after the flash is triggered is attributed to electronic conduction. Meanwhile, the lack of frequency response and insensitiveness to the type of electrical field found in the case of BFO can be attributed to its mainly electronic bulk conduction.
Mayo, 2023 · DOI: 10.1016/j.ceramint.2022.06.242
Nanotecnología en Superficies y Plasma
Surface Acoustic Waves Equip Materials with Active De-Icing Functionality: Unraveled Glaze Ice De-Icing Mechanisms and Application to Centimeter-Scale Transparent Surfaces
Jacob, S; Pandey, S; Del Moral, J; Karimzadeh, A; Gil-Rostra, J; Gonzalez-Elipe, AR; Borras, A; Winkler, AAdvanced Materials Technologies DOI: 10.1002/admt.202300263
Abstract
Enabling active de-icing functionality on low heat conductive and transparent materials is a requirement for several seminal industries in critical economic sectors. However, developing efficient and environmentally friendly de-icing methods still fails because of compatibility problems with large-scale devices and real-world conditions. In this paper, de-icing several square centimeters covered with thick layers of glaze ice is approached through nanoscale activation by surface acoustic waves (SAWs). De-icing functionality is demonstrated with a self-supported piezoelectric material (LiNbO3) and a piezoelectric film (ZnO) deposited on fused silica, the latter system proving the compatibility of the method with materials of practical relevance. Its applicability to large and transparent substrates is demonstrated by placing the interdigitated electrodes (IDTs) required for activation close to the substrate's edges, leaving most of the surface unaltered. The de-icing mechanism of glaze ice by SAW activation is revealed by simulating the SAW propagation on ice-covered surfaces and by experimental analysis of the ice melting process. This involves a combination of ice mechanical stress activation and heating through the initially formed water/ice front. Possible Joule effects due to ohmic losses in the IDTs have been discarded, monitoring local temperature variations during SAW activation at and out of resonance conditions.
Mayo, 2023 · DOI: 10.1002/admt.202300263
Tribología y Protección de Superficies
Tribological Response of delta-Bi2O3 Coatings Deposited by RF Magnetron Sputtering
Rodil, SE; Depablos-Rivera, O; Sanchez-Lopez, JCLubricants, 11 (2023) 207 DOI: 10.3390/lubricants11050207
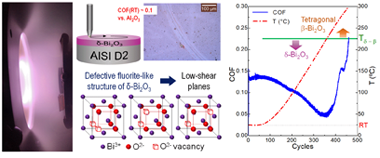
Abstract
Bismuth oxide (Bi2O3) coatings and composite coatings containing this oxide have been studied due to their potential applications in gas sensing, optoelectronics, photocatalysis, and even tribology. Two parametric models based on chemical features have been proposed with the aim of predicting the lubricity response of oxides. However, such models predict contradictory values of the coefficient of friction (COF) for Bi2O3. In this study, we deposited Bi2O3 coatings, via magnetron sputtering, on AISI D2 steel substrates to evaluate the tribological responses of the coatings and determine which parametric model describes them better. Experimentally, only coatings presenting the cubic defective fluorite-like delta-Bi2O3 phase could be evaluated. We performed pin-on-disk tests at room temperature and progressively increasing temperatures up to 300 degrees C using alumina and steel counter-bodies. Low wear and COFs (0.05 to 0.15) indicated that the delta-phase behaves as a lubricious solid, favoring the validity of one of the models. An alternative explanation is proposed for the low COF of the defective fluorite-like structure since it is well known that it contains 25% of anionic vacancies that can be ordered to form low shear-strength planes, similar to the Magneli phases. Two challenges for future potential applications were observed: one was the low adhesion strength to the substrate, and the other was the thermal stability of this phase.
Mayo, 2023 · DOI: 10.3390/lubricants11050207
Nanotecnología en Superficies y Plasma
Strontium/zinc phytate-based self-assembled monolayers on titanium surfaces enhance osteogenesis and antibacterial performance in vitro
Asensio, G; Hernández-Arriaga, AM; Martin-del-Campo, M; Prieto, MA; González-Elipe, AR; Rojo, L; Vázquez-Lasa, BApplied Surface Science, 620 (2023) 156818 DOI: 10.1016/j.apsusc.2023.156818
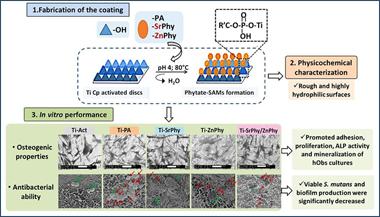
Abstract
The accumulation of bacteria over implant surfaces is still the first cause of failure, and the development of antimicrobial surfaces constitutes a first line in implant research. Besides, the durability and mechanical performance of implants, in special in the dental area, are mainly determined by their osseointegration capacity into the maxillofacial bone and the appearance of infections. Consequently, implant osseointegration and infection prophylaxis remain as big challenges to attain so a huge investigation is being developed on the production of bioactive surfaces to achieve improvements in these aspects. In this work we propose the functionalization of titanium surfaces (Ti Cp) with self-assembled monolayers (SAMs) of bioactive organophosphate compounds: phytic acid (Ti-PA) and its metallic phytate de- rivatives bearing Sr2+ and/or Zn2+ (Ti-SrPhy, Ti-ZnPhy and Ti-SrPhy/ZnPhy) which exhibited tunable in vitro osteogenic, antimicrobial and antioxidant properties in a previous work. Thus, phytate compounds are chemically anchored onto Ti discs through a simple procedure consisting of a condensation reaction promoted by heat treatment. EDS and XPS spectroscopies confirm the obtaining of the modified surfaces and the topographic properties and wettability analysed by SEM, AFM, profilometry and contact angle measurements, respectively, are explored. Additionally, phytate-SAMs do not release any cytotoxic compound after 14 days and stimulate in vitro adhesion and proliferation of human osteoblast cells after 14 days of culture. The osteogenic ability of the modified surfaces evaluated by the quantification of ALP activity and matrix mineralization degree shows a significant improvement with respect to unmodified surfaces. Furthermore, the antimicrobial activity of phytate-SAMs against Streptococcus mutans cultures is evaluated. The count of viable cells and the quantification of produced biofilm are significantly reduced by all phytate-SAMs groups (p < 0.001). Cell membrane integrity studies by LIVE/DEAD staining and SEM imaging confirm a decreased viability of adhered bacteria when phytate-based surfaces are tested, due to a disruption in the function and permeability of the cell membrane. Therefore, phytate-SAMs exhibit suitable in vitro features suggesting their promising potential as bioactive coatings of dental implants.
Mayo, 2023 · DOI: 10.1016/j.apsusc.2023.156818
Reactividad de Sólidos
Direct comparison of surface crystal growth kinetics in chalcogenide glass measured by microscopy and DSC
Shanelova, J; Honcova, P; Malek, J; Perejon, A; Perez-Maqueda, LAJournal of the American Ceramic Society, 106 (2023) 6051-6061 DOI: 10.1111/jace.19204
Abstract
Surface crystallization in fine powder Se70Te30 chalcogenide glass was studied by differential scanning calorimetry (DSC) and optical microscopy. A complex kinetic analysis of these experimental data reveals that the contracting sphere mechanism (R3 model) is the rate determining step of crystal growth, and the conventional Johnson-Mehl-Avrami-Kolmogorov model cannot be used in this case. Moreover, it is clearly shown that the particle size distribution should be considered in crystallization studies. Actually, when the particle size effect is taken into account, the simulated DSC curves for the R3 model agree very well with the experimental data over the entire temperature range. The crystallization kinetics determined from the nonisothermal DSC data are consistent with previously reported isothermal crystallization data for the same powder fraction. The crystal growth rate calculated from isothermal and nonisothermal DSC data agrees very well with the microscopically measured surface and bulk crystal growth rate.
Mayo, 2023 · DOI: 10.1111/jace.19204
Reactividad de Sólidos
Limits of powder metallurgy to fabricate porous Ti35Nb7Zr5Ta samples for cortical bone replacements
Rodriguez-Albelo, LM; Navarro, P; Gotor, FJ; de la Rosa, JE; Mena, D; Garcia-Garcia, FJ; Beltran, AM; Alcudia, A; Torres, YJournal of Materials Research and Technology-JMR&T, 24 (2023) 6212-6226 DOI: 10.1016/j.jmrt.2023.04.212
Abstract
The use of 13-Titanium alloys to fabricate metal implants with Young's modulus that re-sembles bone tissues is presented as an alternative to commercially pure titanium or a- Titanium alloys, although it is still necessary to introduce proper implant porosity to reach the Young's modulus of cortical bones. In this work, porous samples were fabricated by loose sintering (0 MPa) and compared to samples manufactured at 1000 MPa, both sintered under the same conditions. Raw powders and sintered samples of the 13-Titanium alloy, Ti35Nb7Zr5Ta, were characterized in detail in terms of both physicochemical and micro-structural properties. Moreover, the tribo-mechanical behavior of sintered samples was evaluated by performing ultrasound technique, instrumented micro-indentation (P-h curves), and scratch tests. The bio-functional behavior was studied by impedance spec-troscopy and contact angle measurements. The results allowed the evaluation of the limits of conventional powder metallurgy (percentage of porosity, size, and morphology of pores), as well as the influence of the porosity and chemical composition to achieve a better biomechanical and bio-functional behavior that would guarantee bone requirements. The Ti35Nb7Zr5Ta alloy showed relatively high electrical impedance values compared to commercially pure titanium, indicating an improved bio-corrosion behavior. Furthermore, wettability measurements indicated that porous disks fabricated by loose sintering exhibit higher hydrophilicity, often associated with a better antibacterial response
Mayo, 2023 · DOI: 10.1016/j.jmrt.2023.04.212
Nanotecnología en Superficies y Plasma
Advanced Cellulose-Nanocarbon Composite Films for High-Performance Triboelectric and Piezoelectric Nanogenerators
Gonzalez, J; Ghaffarinejad, A; Ivanov, M; Ferreira, P; Vilarinho, PM; Borras, A; Amorin, H; Wicklein, BNanomaterials, 13 (2023) 1206 DOI: 10.3390/nano13071206
Abstract
Natural polymers such as cellulose have interesting tribo- and piezoelectric properties for paper-based energy harvesters, but their low performance in providing sufficient output power is still an impediment to a wider deployment for IoT and other low-power applications. In this study, different types of celluloses were combined with nanosized carbon fillers to investigate their effect on the enhancement of the electrical properties in the final nanogenerator devices. Cellulose pulp (CP), microcrystalline cellulose (MCC) and cellulose nanofibers (CNFs) were blended with carbon black (CB), carbon nanotubes (CNTs) and graphene nanoplatelets (GNPs). The microstructure of the nanocomposite films was characterized by scanning electron and probe microscopies, and the electrical properties were measured macroscopically and at the local scale by piezoresponse force microscopy. The highest generated output voltage in triboelectric mode was obtained from MCC films with CNTs and CB, while the highest piezoelectric voltage was produced in CNF-CNT films. The obtained electrical responses were discussed in relation to the material properties. Analysis of the microscopic response shows that pulp has a higher local piezoelectric d(33) coefficient (145 pC/N) than CNF (14 pC/N), while the macroscopic response is greatly influenced by the excitation mode and the effective orientation of the crystals relative to the mechanical stress. The increased electricity produced from cellulose nanocomposites may lead to more efficient and biodegradable nanogenerators.
Abril, 2023 · DOI: 10.3390/nano13071206
Química de Superficies y Catálisis
Scalable synthesis of 2D Ti2CTx MXene and molybdenum disulfide composites with excellent microwave absorbing performance
Miao, BJ; Cao, YE; Zhu, QS; Nawaz, MA; Ordiozola, JA; Reina, TR; Bai, ZM; Ren, JN; Wei, FCAdvanced Composites and Hybrid Materials, 6 (2023) 61 DOI: 10.1007/s42114-023-00643-2
Abstract
The signal crosstalk and electromagnetic interference (EMI) problems direly need to be resolved in the rapid development of modern microwave communication technology for a better working frequency and transmission power of electronic systems. Where the new absorbing materials such as molybdenum disulfide (MoS2)/titania (TiO2)/Ti2CTx and MoS2/Ti2CTx composites could meet the requirement of "thin, strong, light weight, and wide band" for excellent absorbing performance. In this work, a lighter Ti2CTx material was selected as the matrix, and MoS2 was in-situ grown on Ti2CTx matrix by traditional hydrothermal method and microwave solvothermal method. The fabricated composite exhibited synergic effect of two-dimensional heterostructural interface and double dielectric elements, where a small amount of TiO2 and a certain proportion of MoS2 jointly improve the impedance matching of the composite material. In here, the extreme reflection loss (RLmin) can reach - 54.70 dB (with a frequency of 7.59 GHz, 3.39 mm thickness), and the maximum effective absorption bandwidth (EAB(max)) can reach 4 GHz. Polyethylene glycol 200 was used as the solvent instead of water to make Ti2CTx less oxidized during the composite process, where the microwave heating would attain fast speed, short time, high efficiency, and uniform product. Since, the MoS2/Ti2CTx composite without oxidizing possessed a wider effective absorption bandwidth (EAB) at a thinner thickness, thus resulting in the excellent microwave absorption performance and confirming the validity and rationality of new microwave absorption materials.
Abril, 2023 · DOI: 10.1007/s42114-023-00643-2
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
High-Performance Photocatalytic H2 Production Using a Binary Cu/ TiO2/SrTiO3 Heterojunction
Gonzalez-Tejero, M; Villachica-Llamosas, JG; Ruiz-Aguirre, A; Colon, GACS Applied Energy Materials, 6 (2023) 4007-4015 DOI: 10.1021/acsaem.3c00219
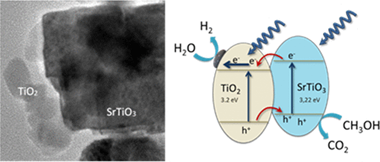
Abstract
Cu/TiO2/SrTiO3 hybrid structures have been synthesized by the simple impregnation method from Cu/TiO2 and SrTiO3 systems. The structural and surface characterization stated that Cu/TiO2/SrTiO3 composites form an effective covering of SrTiO3 by Cu/TiO2. The heterostructured catalysts lead to an outstanding improved photoactivity for hydrogen production from methanol photoreforming that would be related with the efficient separation of charge pairs favored by the Cu/ TiO2/SrTiO3 heterojunction. The best photoproduction is attained for the 30 wt % SrTiO3 heterojunction showing 81.7 mmol/g H2 after 6 h (leading to an apparent quantum yield of ca 1%), 1.7 times higher than that of bare Cu/TiO2.
Abril, 2023 · DOI: 10.1021/acsaem.3c00219
Reactividad de Sólidos
Large-scale oxygen-enriched air (OEA) production from polymeric membranes for partial oxycombustion processes
Garcia-Luna, S; Ortiz, C; Chacartegui, R; Perez-Maqueda, LAEnergy, 268 (2023) 126697 DOI: 10.1016/j.energy.2023.126697
Abstract
Partial oxycombustion using Oxygen-Enriched Air (OEA), produced by air-gas separation with polymeric membranes, combined synergistically with CO2 capture technologies, can reduce the overall energy cost of CO2 capture, and it is a potential alternative to conventional CO2 capture technologies. An exhaustive review of polymeric membranes for this application is presented. The best membranes showed permeability values in the 450-25,100 barrer and selectivities higher than 3.6 for large-scale operations. These membranes can produce OEA with oxygen molar concentrations of up to 40% for retrofitting large-scale power plants (similar to 500 MWe) with partial oxycombustion. For OEA production, the polymeric membrane system is more efficient than cryogenic distillation since the specific power consumption of the former is 35.17 kWh/ton OEA. In comparison, that of the latter is 49.57 kWh/ton OEA. This work proposes that the OEA produced by the membranes feed a partial oxycombustion process integrated with calcium looping within a hybrid CO2 capture system. The power con-sumption of the hybrid CO2 capture system proposed here is 29.05% lower than in the case OEA is produced from cryogenic distillation, which justifies the potential interest in using polymeric membranes for OEA production.
Abril, 2023 · DOI: 10.1016/j.energy.2023.126697
Fotocatálisis Heterogénea: Aplicaciones
Photocatalytic treatment based on TiO2 for a coal mining drainage
Murcia-Mesa, JJ; Patino-Castillo, CG; Rojas-Sarmiento, HA; Navio-Santos, A; Hidalgo-Lopez, MD; Angel-Botero, ARevista Facultad de Ingeniería-Universidad de Antioquia, 107 (2023) 88-101 DOI: 10.17533/udea.redin.20211063
Abstract
The aim of the present work was to evaluate the effectiveness of a heterogeneous photocatalyst based on TiO2 in the treatment of coal mining drainage which contains a variety of heavy metals and high concentration sulfates and sulfides. The photocatalytic behavior of the commercial reference Sigma Aldrich and the different materials synthesized using the Sol-gel methodology with surface modifications using sulfation and fluorination processes were analyzed. To find a possible correlation between the physicochemical properties of photocatalysts and their behavior, a characterization was carried out using X-Ray Diffraction (XRD), X-Ray Fluorescence spectrometry (XRF), Fourier transform infrared spectroscopy (FT-IR), UV-Vis diffuse reflectance Spectra (UV-Vis DRS), N2 physisorption, X-ray photoelectron spectroscopy (XPS), and particle size analysis. Results indicated that the modification of the TiO2 prepared in the laboratory using sulfation and fluorination allowed the successful control of the physicochemical properties of this oxide. However, commercial TiO2 showed the greatest effectiveness in removing metals such as: Fe, Cu, Cr, and As after a photocatalytic reaction for a maximum of 1 hour under continuous nitrogen flow and a light intensity of 120 W/m2.
Abril, 2023 · DOI: 10.17533/udea.redin.20211063
Química de Superficies y Catálisis
In-situ DRIFTS steady-state study of CO2 and CO methanation over Ni-promoted catalysts
González-Castaño, M; González-Arias, J; Bobadilla, LF; Ruíz-López, E; Odriozola, JA; Arellano-García, HFuel, 338 (2023) 127241 DOI: 10.1016/j.fuel.2022.127241
Abstract
Promoting the performance of catalytic systems by incorporating small amount of alkali has been proved effective for several reactions whilst controversial outcomes are reported for the synthetic natural gas production. This work studies a series of Ni catalysts for CO2 and CO methanation reactions. In-situ DRIFTS spectroscopy evidenced similar reaction intermediates for all evaluated systems and it is proposed a reaction mechanism based on: i) formate decomposition and ii) hydrogenation of lineal carbonyl species to methane. Compared to bare Ni, the enhanced CO2 methanation rates attained by NiFe/Al and NiFeK/Al systems are associated to promoted formates decomposition into lineal carbonyl species. Also for CO methanation, the differences in the catalysts' performances were associated to the relative concentration of lineal carbonyl species. Under CO methanation conditions and opposing the CO2 methanation results where the incorporation of K delivered promoted catalytic behaviours, worsened CO methanation rates were discerned for the NiFeK/Al system.
Abril, 2023 · DOI: 10.1016/j.fuel.2022.127241
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Facile Synthesis of Heterogeneous Indium Nanoparticles for Formate Production via CO2 Electroreduction
Perez-Sequera, AC; Diaz-Perez, MA; Angulo, MAL; Holgado, JP; Serrano-Ruiz, JCNanomaterials, 13 (2023) 3052 DOI: 10.3390/nano13081304
Abstract
In this study, a simple and scalable method to obtain heterogeneous indium nanoparticles and carbon-supported indium nanoparticles under mild conditions is described. Physicochemical characterization by X-ray diffraction (XRD), X-ray photoelectron microscopy (XPS), scanning electron microscopy (SEM) and transmission electron microscopy (TEM) revealed heterogeneous morphologies for the In nanoparticles in all cases. Apart from In-0, XPS revealed the presence of oxidized In species on the carbon-supported samples, whereas these species were not observed for the unsupported samples. The best-in-class catalyst (In-50/C-50) exhibited a high formate Faradaic efficiency (FE) near the unit (above 97%) at -1.6 V vs. Ag/AgCl, achieving a stable current density around -10 mA center dot cm(geo)(-2), in a common H-cell. While In-0 sites are the main active sites for the reaction, the presence of oxidized In species could play a role in the improved performance of the supported samples.
Abril, 2023 · DOI: 10.3390/nano13081304
Reactividad de Sólidos
Electrical performance of orthotropic and isotropic 3YTZP composites with graphene fillers
Lopez-Pernia, C; Muñoz-Ferreiro, C; Moriche, R; Morales-Rodriguez, A; Gallardo-Lopez, A; Poyato, RJournal of The European Ceramic Society, 43 (2023) 1605-1612 DOI: 10.1016/j.jeurceramsoc.2022.11.068
Abstract
3 mol% yttria tetragonal zirconia polycrystal (3YTZP) composites with orthotropic or isotropic microstructures were obtained incorporating few layer graphene (FLG) or exfoliated graphene nanoplatelets (e-GNP) as fillers. Electrical conductivity was studied in a wide range of contents in two configurations: perpendicular (sigma(perpendicular to)) and parallel (sigma(//)) to the pressing axis during spark plasma sintering (SPS). Isotropic e-GNP composites presented excellent electrical conductivity for high e-GNP contents (sigma(perpendicular to)similar to 3200 S/m and sigma(//) similar to 1900 S/m for 20 vol% e-GNP), consequence of their misoriented distribution throughout the matrix. Optimum electrical performance was achieved in the highly anisotropic FLG composites, with high electrical conductivity for low contents (sigma(perpendicular to) similar to 680 S/m for 5 vol%), percolation threshold below 2.5 vol% FLG and outstanding electrical conductivity for high contents (sigma(perpendicular to) similar to 4000 S/m for 20 vol%), result of the high aspect ratio and low thickness of FLG.
Abril, 2023 · DOI: 10.1016/j.jeurceramsoc.2022.11.068
Materiales de Diseño para la Energía y Medioambiente
The Role of Protective Surface Coatings on the Thermal Stability of Delithiated Ni-Rich Layered Oxide Cathode Materials
Reissig, F; Ramirez-Rico, J; Placke, TJ; Winter, M; Schmuch, R; Gomez-Martin, ABatteries-Basel, 9 (2023) 245 DOI: 10.3390/batteries9050245
Abstract
To achieve a broader public acceptance for electric vehicles based on lithium-ion battery (LIB) technology, long driving ranges, low cost, and high safety are needed. A promising pathway to address these key parameters lies in the further improvement of Ni-rich cathode materials for LIB cells. Despite the higher achieved capacities and thus energy densities, there are major drawbacks in terms of capacity retention and thermal stability (of the charged cathode) which are crucial for customer acceptance and can be mitigated by protecting cathode particles. We studied the impact of surface modifications on cycle life and thermal stability of LiNi0.90Co0.05Mn0.05O2 layered oxide cathodes with WO3 by a simple sol-gel coating process. Several advanced analytical techniques such as low-energy ion scattering, differential scanning calorimetry, and high-temperature synchrotron X-ray powder diffraction of delithiated cathode materials, as well as charge/discharge cycling give significant insights into the impact of surface coverage of the coatings on mitigating degradation mechanisms. The results show that successful surface modifications of WO3 with a surface coverage of only 20% can prolong the cycle life of an LIB cell and play a crucial role in improving the thermal stability and, hence, the safety of LIBs.
Abril, 2023 · DOI: 10.3390/batteries9050245
Materiales Ópticos Multifuncionales - Materiales Coloidales
Oxidation and coordination states assumed by transition metal dopants in an invert ultrabasic silicate glass
Zandona, A; Castaing, V; Shames, AI; Helsch, G; Deubener, J; Becerro, AI; Allix, M; Goldstein, AJournal of Non-Crystalline Solids, 603 (2023) 122094 DOI: 10.1016/j.jnoncrysol.2022.122094
Abstract
An ultrabasic invert silicate glass (46SiO2.11Na2O.21CaO.22BaO, optical basicity index equal to 0.71) was synthesized (O2 atmosphere) and used as host for various transition metal dopants. Optical absorption, emission and electron paramagnetic spectroscopies were used to characterize oxidation and coordination states. Some of the dopants displayed only their maximal oxidation state (Ti4+, V5+, Cr6+, Mo6+ and W6+). Others exhibited mixed valences: (i) Mn3+ was the dominant species, alongside Mn2+ and Mn5+; (ii) stable Fe3+ prevailed, although some Fe2+ was preliminarily suggested by the absorption spectrum; (iii) Co3+ probably accompanied the dominant Co2+ tetrahedral oxide complex; (iv) like in "conventional" silicate glasses, only Ni2+ was detected, though simultaneously located in tetrahedral and octahedral sites (somewhat distorted); (v) Cu+ was surprisingly identified alongside the expected 6-fold coordinated Cu2+. Drastic reduction of the oxygen content in the melting atmosphere led to conversion of Cr6+ to Cr3+, despite the extreme basicity of the host.
Marzo, 2023 · DOI: 10.1016/j.jnoncrysol.2022.122094
Química de Superficies y Catálisis
Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production
Bounoukta, CE; Megias-Sayago, C; Navarro, JC; Ammari, F; Ivanova, S; Centeno, MANanomaterials, 13 (2023) 1129 DOI: 10.3390/nano13061129
Abstract
Cotton stalks-based biochars were prepared and used to synthetize Ru-supported catalysts for selective production of gamma-valerolactone from levulinic acid in aqueous media. Different biochars' pre-treatments (HNO3, ZnCl2, CO2 or a combination of them) were carried out to activate the final carbonaceous support. Nitric acid treatment resulted in microporous biochars with high surface area, whereas the chemical activation with ZnCl2 substantially increases the mesoporous surface. The combination of both treatments led to a support with exceptional textural properties allowing the preparation of Ru/C catalyst with 1422 m(2)/g surface area, 1210 m(2)/g of it being a mesoporous surface. The impact of the biochars' pre-treatments on the catalytic performance of Ru-based catalysts is fully discussed.
Marzo, 2023 · DOI: 10.3390/nano13061129
Nanotecnología en Superficies y Plasma
Microstructural Characterization and Self-Propagation Properties of Reactive Al/Ni Multilayers Deposited onto Wavelike Surface Morphologies: Influence on the Propagation Front Velocity
Camposano, YHS; Bartsch, H; Matthes, S; Oliva-Ramirez, M; Jaekel, K; Schaaf, PPhysica Status Solidi A-Applications and Materials Science (2023) 2200765 DOI: 10.1002/pssa.202200765
Abstract
Reactive multilayer systems are nanostructures of great interest for various technological applications because of their high energy release rate during the self-propagating reaction of their components. Therefore, many efforts are aimed at controlling the propagation velocity of these reactions. Herein, reactive multilayer systems of Al/Ni in the shape of free-standing foils with a wavelike surface morphology prepared by using sacrificial substrates with well-aligned waves are presented and the propagation of the reaction along different directions of the reproduced waves is analyzed. During the ignition test, the propagation front is recorded with a high-speed camera, and the maximum temperature is measured using a pyrometer. The propagation of the reaction is favored in the direction of the waves, which points out the influence of the anisotropy generated by this morphology and how it affects the propagation dynamics and the resulting microstructure. Furthermore, compared to their counterparts fabricated on flat substrates, these reactive multilayers with wavelike morphology exhibit a remarkable reduction in the propagation velocity of the reaction of about 50%, without significantly affecting the maximum temperature registered during the reaction.
Marzo, 2023 · DOI: 10.1002/pssa.202200765
Materiales y Procesos Catalíticos de Interés Ambiental y Energético
Revealing the Impact of Different Iron-Based Precursors on the ‘Catalytic’ Graphitization for Synthesis of Anode Materials for Lithium Ion Batteries
Frankenstein, L; Glomb, P; Ramirez-Rico, J; Winter, M; Placke, T; Gomez-Martin, AChemelectrochem, 10 (2023) e202201073 DOI: 10.1002/celc.202201073
Abstract
Low cost and environmentally friendly production of graphite anodes from naturally available biomass resources is of great importance to satisfy the increasing material demand for lithium ion batteries. Herein, graphitization of coffee ground was performed using four different iron-based activating additives, including iron (III) chloride, iron (III) nitrate, iron (III) oxide and pure iron, following either a wet or a dry mixing approach. The structural development regarding the type of activator used and the impact on the corresponding electrochemical performance are systematically investigated. A maximum degree of graphitization between 55 and 74 % (as determined by Raman spectroscopy) is attained using iron (III) chloride and iron powder, respectively. The graphitic anode material synthesized using iron powder reached a maximum reversible capacity of approximate to 320 mAh g(-1) at a rate of 0.1 C. This study provides significant insights into the impact of activators on the design of synthetic graphite from renewable sources.
Marzo, 2023 · DOI: 10.1002/celc.202201073
Nanotecnología en Superficies y Plasma
Improved strain engineering of 2D materials by adamantane plasma polymer encapsulation
Carrascoso, F; Li, H; Obrero-Perez, JM; Aparicio, FJ; Borras, A; Island, JO; Barranco, A; Castellanos-Gómez, ANPJ 2D Materials and Applications, 7 (2023) 24 DOI: 10.1038/s41699-023-00393-1
Abstract
Two-dimensional materials present exceptional crystal elasticity and provide an ideal platform to tune electrical and optical properties through the application of strain. Here we extend recent research on strain engineering in monolayer molybdenum disulfide using an adamantane plasma polymer pinning layer to achieve unprecedented crystal strains of 2.8%. Using micro-reflectance spectroscopy, we report maximum strain gauge factors of -99.5 meV/% and -63.5 meV/% for the A and B exciton of monolayer MoS2, respectively, with a 50 nm adamantane capping layer. These results are corroborated with photoluminescence and Raman measurements on the same samples. Taken together, our results indicate that adamantane polymer is an exceptional capping layer to transfer substrate-induced strain to a 2D layer and achieve higher levels of crystal strain.
Marzo, 2023 · DOI: 10.1038/s41699-023-00393-1
Materiales Ópticos Multifuncionales
Origin of anomalously stabilizing ice layers on methane gas hydrates near rock surface
Li, Y; Corkery, RW; Carretero-Palacios, S; Berland, K; Esteso, V; Fiedler, J; Milton, KA; Brevik, I; Bostrom, MPhysical Chemistry Chemical Physics, 25 (2023) 6636-6652 DOI: 10.1039/d2cp04883c
Abstract
Gas hydrates (GHs) in water close to freezing temperatures can be stabilised via the formation of ice layers. In a recent work [Bostrom et al., Astron. Astrophys., A54, 650, 2021], it was found that a surface region with partial gas dilution could be essential for obtaining nano- to micron-sized anomalously stabilizing ice layers. In this paper, it is demonstrated that the Casimir-Lifshitz free energy in multi-layer systems could induce thinner, but more stable, ice layers in cavities than those found for gas hydrates in a large reservoir of cold water. The thickness and stability of such ice layers in a pore filled with cold water could influence the leakage of gas molecules. Additional contributions, e.g. from salt-induced stresses, can also be of importance, and are briefly discussed.
Marzo, 2023 · DOI: 10.1039/d2cp04883c
- ‹ anterior
- 5 of 37
- siguiente ›














