Artículos SCI
2024
2024
Tribología y Protección de Superficies
Synthesis and Characterization of Multilayered CrAlN/Al2O3 Tandem Coating Using HiPIMS for Solar Selective Applications at High Temperature
Sánchez-Pérez, M; Rojas, TR; Reyes, DF; Ferrer, FJ; Farchado, M; Morales, A; Escobar-Galindo, R; Sánchez-López, JCACS Applied Energy Materials, 7 (2024) 438-449
Show abstract ▽
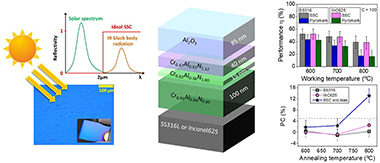
The effect of applying a negative bias during deposition of a previously designed multilayer solar selective absorber coating was studied on two types of substrates (316L stainless steel and Inconel 625). The solar selective coating is composed of different chromium aluminum nitride layers deposited using a combination of radiofrequency (RF), direct current (DC), and high-power impulse magnetron sputtering (HiPIMS) technologies. The chemical composition is varied to generate an infrared reflective/absorber layer (with low Al addition and N vacancies) and two CrAlN intermediate layers with medium and high aluminum content (Al/Cr = 0.6 and 1.2). A top aluminum oxide layer (Al2O3) is deposited as an antireflective layer. In this work, a simultaneous DC-pulsed bias (−100 V, 250 kHz) was applied to the substrates in order to increase the film density. The optical performance, thermal stability, and oxidation resistance was evaluated and compared with the performance obtained with similar unbiased coating and a commercial Pyromark paint reference at 600, 700, and 800 °C. The coating remained stable after 200 h of annealing at 600 °C, with solar absorptance (α) values of 93% and 92% for samples deposited on stainless steel and Inconel, respectively, and a thermal emittance ε25°C of 18%. The introduction of additional ion bombardment during film growth through bias assistance resulted in increased durability, thermal stability, and working temperature limits compared with unbiased coatings. The solar-to-mechanical energy conversion efficiency at 800 °C was found to be up to 2 times higher than Pyromark at C = 100 and comparable at C = 1000.
Febrero, 2024 | DOI: 10.1021/acsaem.3c02310
Fotocatálisis Heterogénea: Aplicaciones
Photocatalytic activity enhancement by noble metal deposition on faceted F-TiO2 synthesised by microwave assisted method. A study of selective oxidation of gas-phase ethanol in a FBPR reactor
Hernández-Laverde, M; Murcia, JJ; Morante, N; Sannino, D; Vaiano, V; Navío, JA; Hidalgo, MCCatalysis Today, 433 (2024) 114645
Show abstract ▽
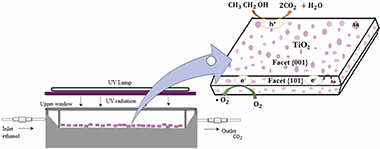
In the present work, fluorinated titanium dioxide (TiO2-F) with high exposition of facet {001} was prepared by following a facile and high yield hydrothermal method assisted by microwave. This faceted TiO2 was then modified by Au or Ag deposition at two different metal loadings (0.125 and 0.25 wt%). A wide physicochemical characterisation of the materials was performed. X-ray difractograms showed high {001} facet exposition in all materials. By X-ray fluorescence it was found that the different samples contained about 5% of fluor. All samples presented high surface area and high uniformity and homogeneity of particles, which highlights the good properties that can be achieved by the microwave synthesis method compared to conventional hydrothermal methods. Oxidation state of the noble metals was studied by XPS. On the other side, TiO2-F and the metallised titania powders were immobilised on polystyrene pellets (PS) for evaluating their gas photocatalytic activity in volatile organic compounds (VOCs) decontamination by following the reaction of photoxidation of ethanol in gas phase. It was found that activity was considerably improved by the addition of noble metals, obtaining high conversion and selectivity to CO2. It is remarkable that the selectivity to CO is almost zero. The highest efficiency was found for the faceted TiO2-F sample with the lowest Au loading (0.125 wt%) immobilised on PS where 91% ethanol conversion and 100% CO2 selectivity were achieved. Different reaction variables were also studied.
Mayo, 2024 | DOI: 10.1016/j.cattod.2024.114645
Química de Superficies y Catálisis
Optimized electrocatalytic degradation of ciprofloxacin using Co3O4 coated stainless steel electrodes
Saleem, MU; Jawad, M; Azad, F; Nawaz, MA; Zaman, WQ; Miran, WColloids and Surfaces A-Physicochemical and Engineering Aspects, 681 (2024) 132738
Show abstract ▽
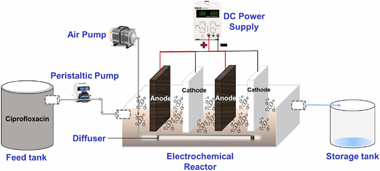
Ciprofloxacin (CIP) is a fluoroquinolone antibiotic that is widely used across the globe and its release is a serious concern due to its persistent nature, partial degradation, and simple transport through different environmental matrices. Pharmaceuticals have been degraded effectively by electrochemical oxidation. Exploring ways to in-crease the mineralization of these compounds while maintaining low power consumption is important. In this study, the treatability and degradation of CIP were investigated by using cobalt oxide-coated stainless steel (SS) electrodes in a lab-scale electrochemical (EC) reactor. The performance of the electrochemical reactor was determined under various operational conditions. The feed wastewater was synthetically prepared in the laboratory with varying concentrations of CIP ranging from 8 to 41 mg/L and the EC reactor was operated with an applied voltage and airflow rate of 2.6-9.3 volts and 1.6-3.5 L/min, respectively. A 3-factor central composite experimental design (CCD) was developed by using response surface methodology (RSM) in Design-Expert software. At a residence time of 27 min, initial concentration of 25 mg/L, airflow rate of 2.5 L/min, and applied voltage of 6 volts, the EC reactor achieved a removal efficiency of 70.8% for CIP with SS electrodes. On the contrary, the removal efficiency was increased to 91.5% at a reduced residence time of 21 min with cobalt oxide (Co3O4) coated over SS plates. The results indicated that Co3O4@SS electrodes resulted in better removal efficiency of CIP at a lower residence time. This system can be used as a robust benchmark for a single or consortium of antibiotics present in domestic and hospital wastewater.
Enero, 2024 | DOI: 10.1016/j.colsurfa.2023.132738
Química de Superficies y Catálisis
Mechanistic insights into methanol carbonylation to methyl acetate over an efficient organic template-free Cu-exchanged mordenite
Luque-Alvarez, LA; Gonzalez-Arias, J; Romero-Sarria, F; Reina, TR; Bobadilla, LF; Odriozola, JACatalysis Science & Technology, 14 (2024) 128-136
Show abstract ▽
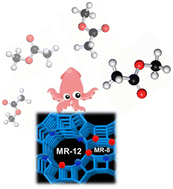
Currently, acetic acid is produced via the carbonylation reaction of methanol with the main route entailing the use of homogeneous noble metal-based catalysts, which has certain limitations, including the use of polluting alkyl halide promoters and difficulty in catalyst recovery. To overcome these challenges, the exploration of alternative methods utilizing heterogeneous catalysts, particularly zeolites with copper as a redox center, has gained attention. Nonetheless, the conversion and selectivity obtained are sought after to compete against the homogeneous route. Therefore, a deeper understanding of the reaction and mechanism is needed to determine the weak points and overcome them. In this study, we propose the use of time-resolved operando DRIFTS-MS to study the methanol carbonylation reaction over a Cu–H-MOR catalyst. The study aimed to propose a reaction mechanism through an investigation of the behavior of the catalyst, including potential identification of the location of the copper redox center in the zeolite. The catalytic performance of the Cu–H-MOR catalyst was also evaluated, demonstrating its activity and stability in the methanol carbonylation reaction. The operando DRIFTS-MS results provide insights into the reaction mechanism and the involvement of the acid and redox centers in the process. Based on the findings, we propose a reaction mechanism for methanol carbonylation on Cu–H-MOR zeolite: (i) methanol dehydration, (ii) CO insertion into methoxide groups, (iii) reaction between dimethyl ether and acetyl groups, and (iv) hydrolysis of methyl acetate. Overall, we believe that this work contributes to a deeper understanding of the heterogeneous route for acetic acid production and offers potential avenues for optimizing the process.
Enero, 2024 | DOI: 10.1039/d3cy01271a
Fotocatálisis Heterogénea: Aplicaciones
Ba3(PO4)2 Photocatalyst for Efficient Photocatalytic Application
Naciri, Y; Ahdour, A; Benhsina, E; Hamza, MA; Bouziani, A; Hsini, A; Bakiz, B; Navio, JA; Ghazzal, MNGlobal Challenges
Show abstract ▽
Barium phosphate (Ba-3(PO4)(2)) is a class of material that has attracted significant attention thanks to its chemical stability and versatility. However, the use of Ba-3(PO4)(2) as a photocatalyst is scarcely reported, and its use as a photocatalyst has yet to be reported. Herein, Ba-3(PO4)(2) nanoflakes synthesis is optimized using sol-gel and hydrothermal methods. The as-prepared Ba-3(PO4)(2) powders are investigated using physicochemical characterizations, including XRD, SEM, EDX, FTIR, DRS, J-t, LSV, Mott-Schottky, and EIS. In addition, DFT calculations are performed to investigate the band structure. The oxidation capability of the photocatalysts is investigated depending on the synthesis method using rhodamine B (RhB) as a pollutant model. Both Ba-3(PO4)(2) samples prepared by the sol-gel and hydrothermal methods display high RhB photodegradation of 79% and 68%, respectively. The Ba-3(PO4)(2) obtained using the sol-gel process exhibits much higher stability under light excitation after four regeneration cycles. The photocatalytic oxidation mechanism is proposed based on the active species trapping experiments where O-2(center dot-) is the most reactive species. The finding shows the promising potential of Ba-3(PO4)(2) photocatalysts and opens the door for further investigation and application in various photocatalytic applications.
Enero, 2024 | DOI: 10.1002/gch2.202300257
- ‹ anterior
- 5 of 410
- siguiente ›
icms











